anti-Perilipin 4 (N-terminus) guinea pig polyclonal, serum
- Guinea pig polyclonal
- Suitable for ICC/IF, IHC and WB
- Reacts with human
Product description
| Quantity | 100 µl |
|---|---|
| Antibody Type | Polyclonal |
| Host | Guinea pig |
| Conjugate | Unconjugated |
| Application | ICC/IF, IHC, WB |
| Purification | Stabilized antiserum |
| Reactivity | Human |
| Storage | Short term at 2-8°C; long term storage in aliquots at -20°C; avoid freeze/thaw cycles |
| Intended use | Research use only |
| Immunogen | Synthetic peptide (N-terminal aa 1-15 of human S3-12; MQLGSFFGSLPGFSS-C, coupled to KLH) |
| Formulation | Contains 0.09% sodium azide and 0.5% BSA |
| UniprotID | Q96Q06 (Human) |
| Synonym | Perilipin-4, Adipocyte protein S3-12, PLIN4, KIAA1881 |
| Note | Centrifuge prior to opening |
Applications
| Tested applications | Tested dilutions |
|---|---|
| Immunocytochemistry (ICC)/ Immunofluorescence (IF) | Assay dependent |
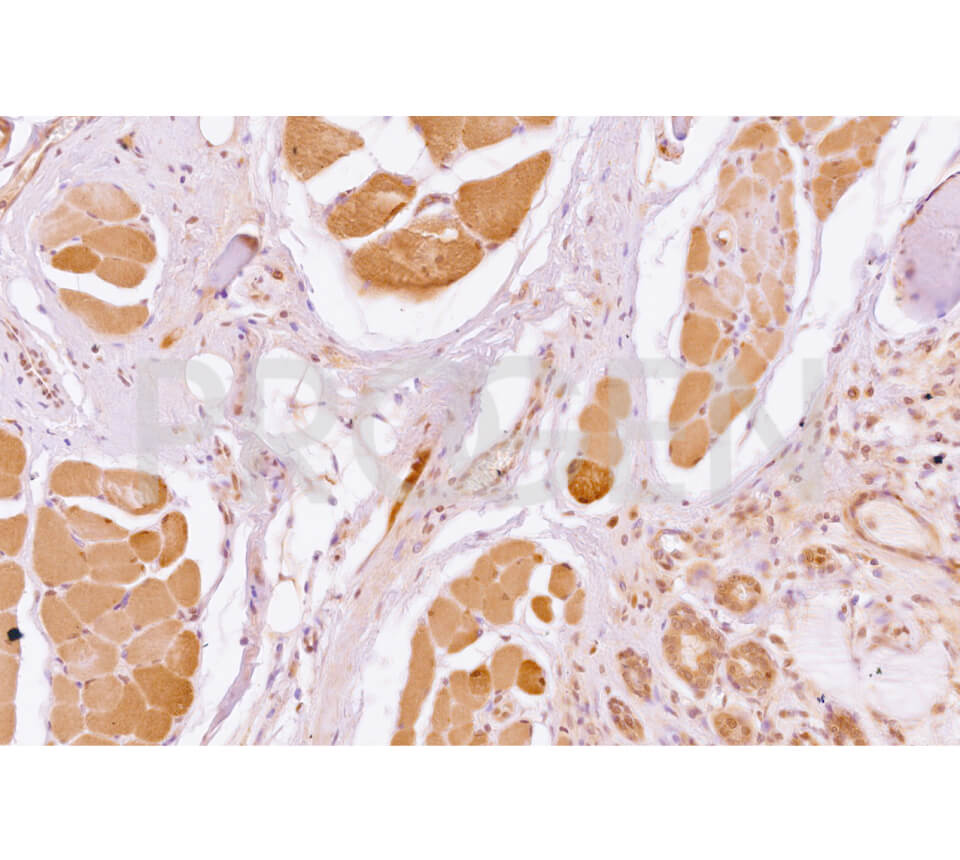
| Immunohistochemistry (IHC) - frozen | 1:100-1:200 |
| Immunohistochemistry (IHC) - paraffin | 1:100-1:200 (microwave treatment recommended) |
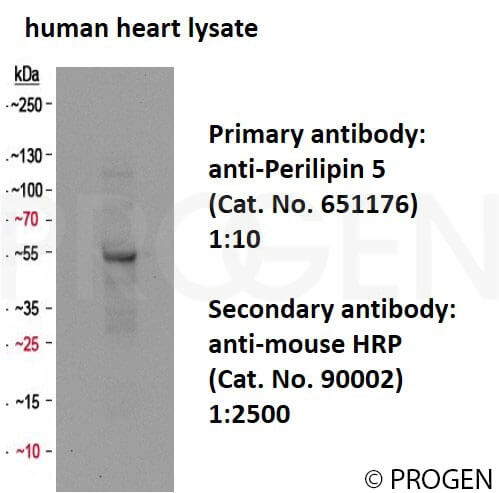
| Western Blot (WB) | 1:2,000 |
Background
Polypeptide reacting: S3-12 (also named PLIN4), apparent Mr 140,000 (after SDS-PAGE).
S3-12 pertains to the PLIN (Perilipin)/PAT-family proteins, covering the surface of cytoplasmic lipid droplets.
Additional PAT proteins include adipophilin (ADRP, perilipin 2), TIP47 (perilipin 3), and MLDP (OXPAT/PAT-1 or LSDP5, or perilipin 5) which are expressed in differentiation-related stages of lipid metabolism.
No cross-reactivity with additional PLIN/PAT proteins (including adipophilin/ADRP/PLIN2, perilipin/PLIN1, and TIP47/PLIN3) which are expressed in differentiation-related stages of lipid metabolism.
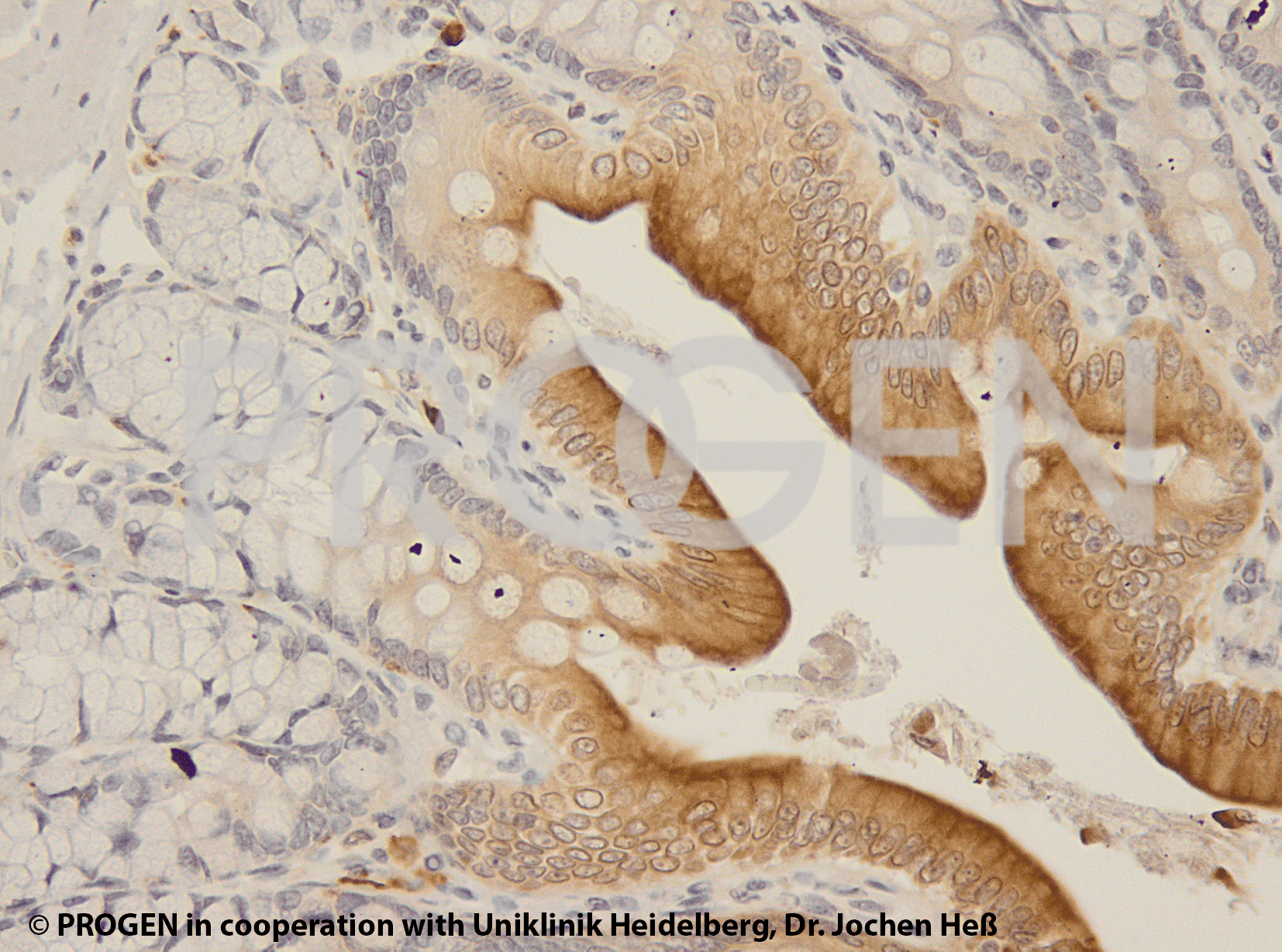
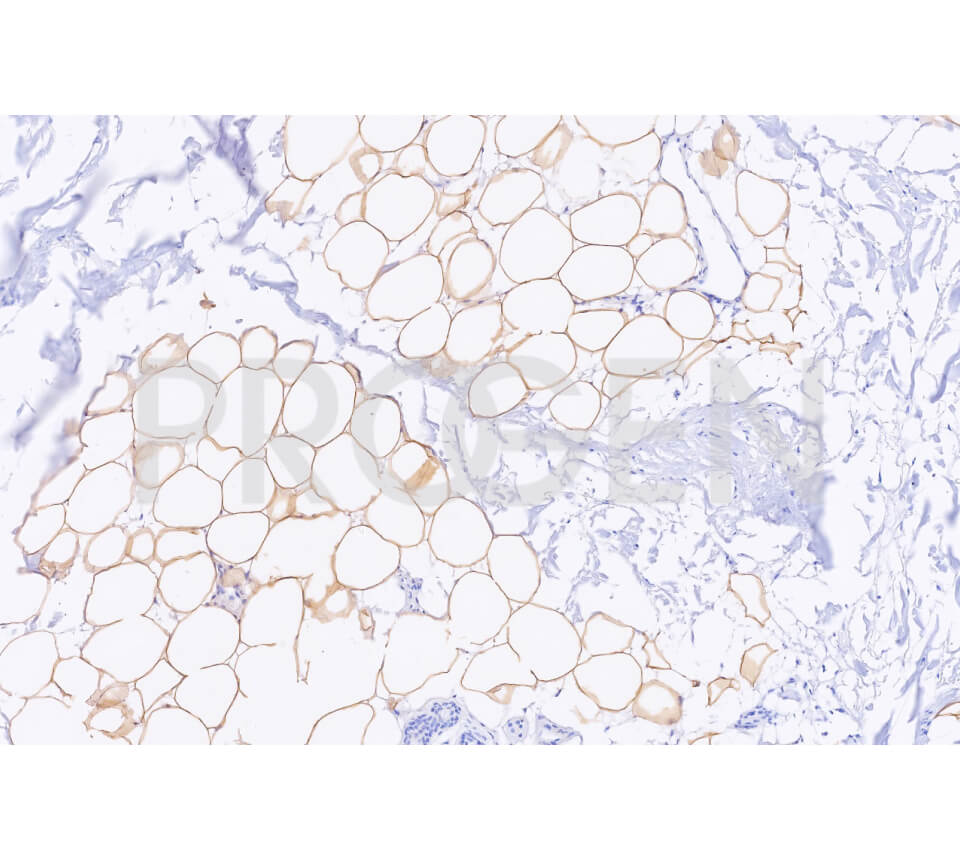
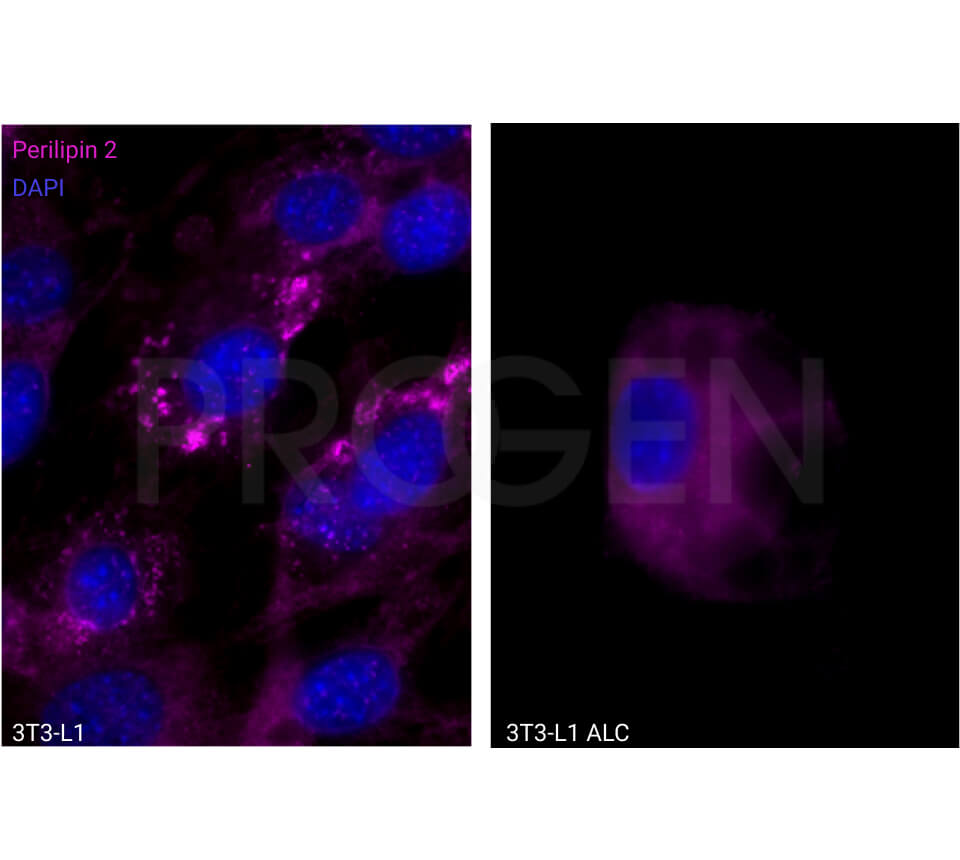
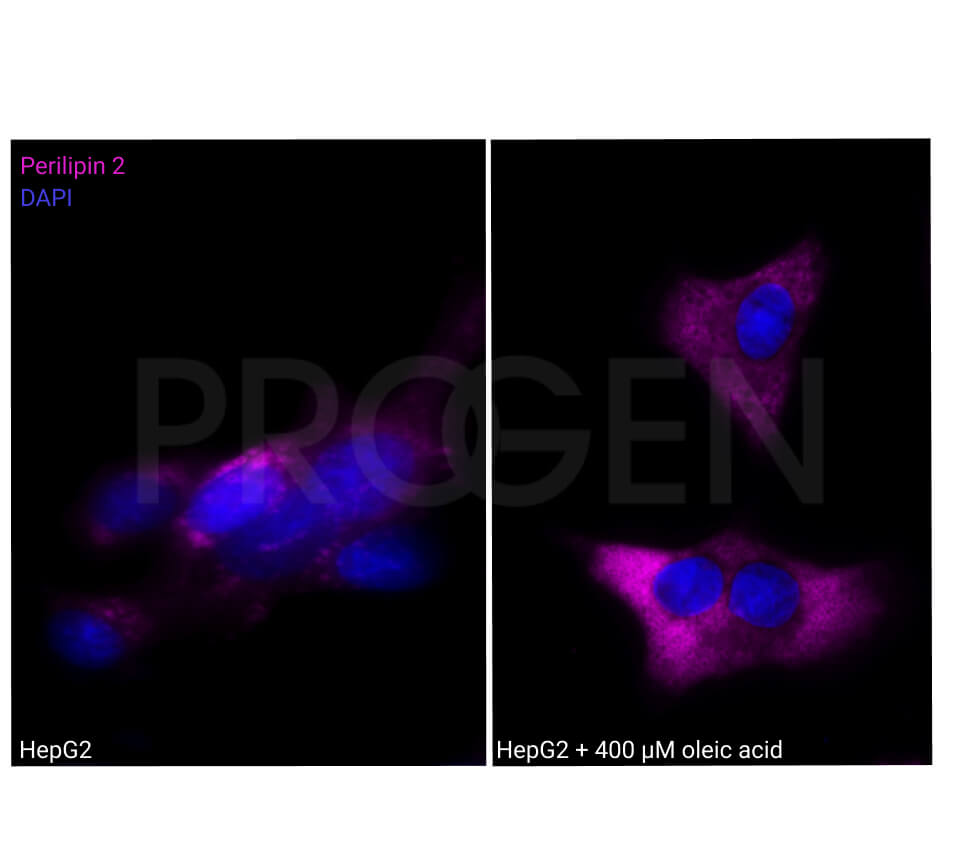
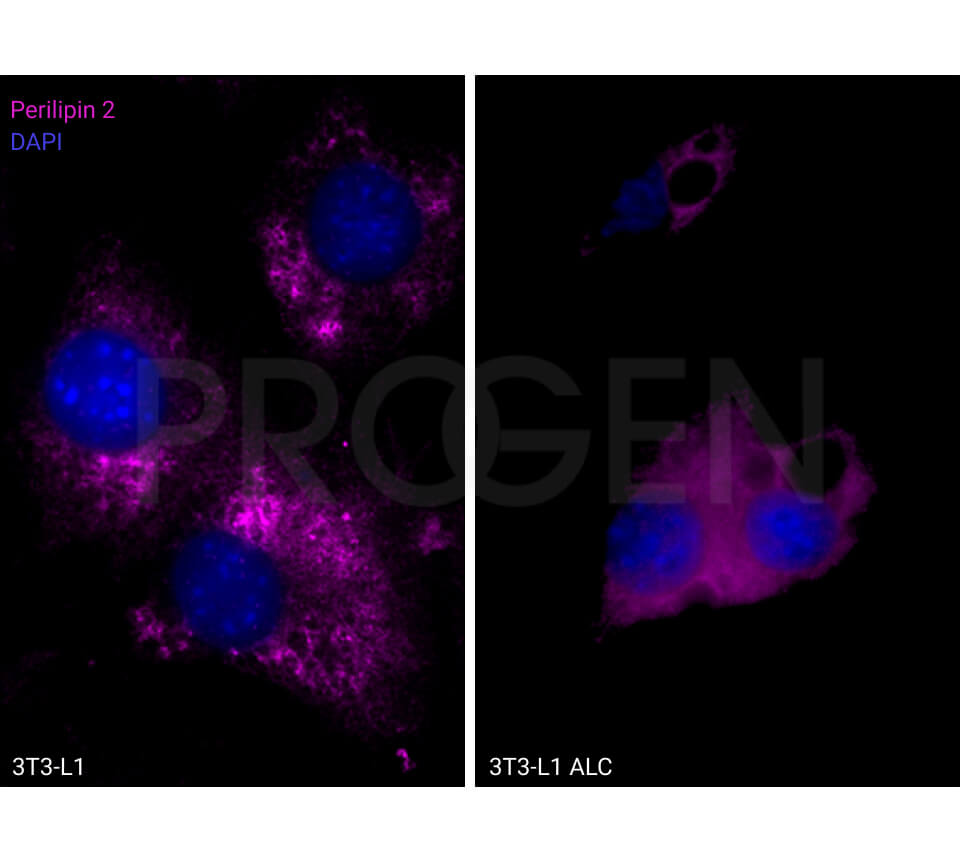
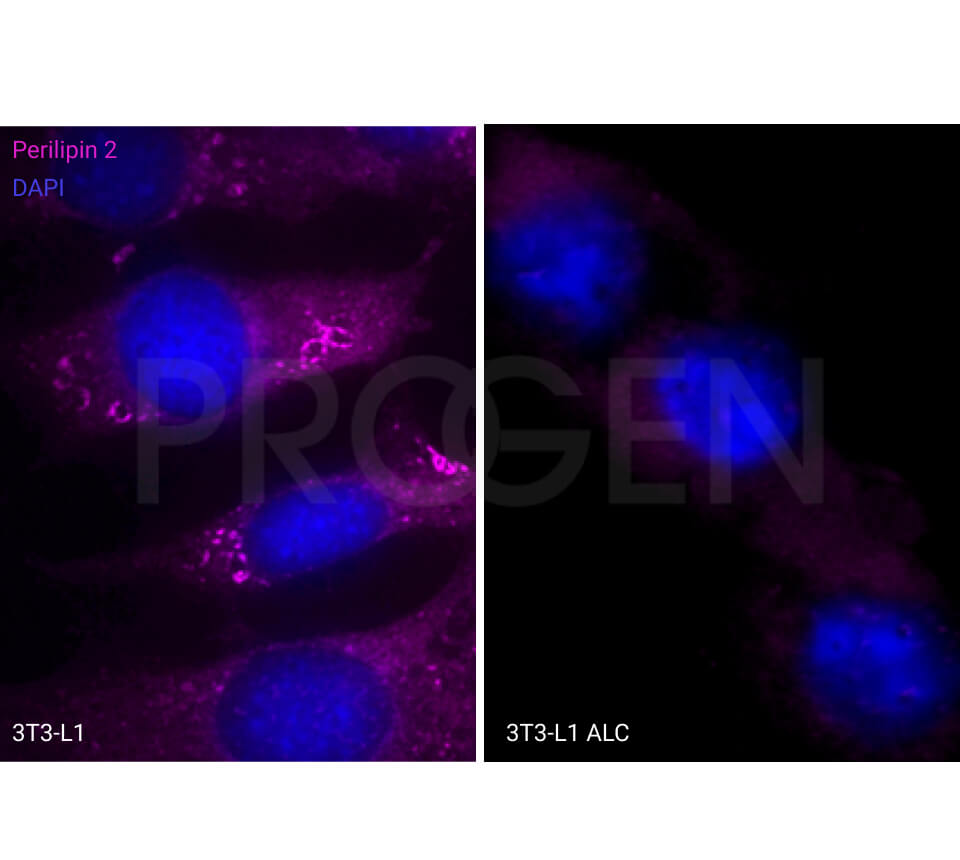
Tissue Immunolocalization: S3-12 is positively detected in lipid droplets of cultured cells.
Reactivities on cultured cell lines: PLC (human).
References/Publications (2)
Downloads
Q & A's
Customer Reviews
Login
FAQs
- PVDF membranes show better results than nitrocellulose (higher capacity, allows for more stringent washing conditions in case of background problems).
- Use freshly prepared blocking solution (e.g. 5% nonfat dry milk, 0.05% Tween 20), block for at least 1 h at room temperature.
- Use the antibody in a higher dilution, but prolong incubation time and exposure time.
- Always use a fresh aliquot of the antibody.
- Do not repeatedly freeze the antibody (eventually centrifuge shortly after thawing to remove cryo-precipitates).
- Include an additional washing step.
You might also try more stringent wash conditions, e.g. add 0.5 M NaCl to the wash buffer. - Always use a fresh aliquot of secondary antibody.
- In case you use ECL most the guinea pig antibody should be diluted further in order to get rid of the background.
The supernatant format contains FCS proteins from cell culture medium supplemented with FCS.
The serum antibodies contain other proteins present in serum.
Most of our liquid antibodies and reconstituted lyophilized antibodies may be stored for short term storage (up to 3 month) at 2-8°C. For long term storage we recommend to store the antibody at -20°C in aliquots. Please avoid freeze and thaw cycles.
Most of our conjugated antibodies should be stored at 2-8°C.
The individual storage conditions are mentioned on the datasheet.
- Methanol/ acetone fixation: Immerse slide in precooled (-20°C) methanol for 5 min, immerse in precooled (-20°C) acetone for 30-60 sec, let specimen air dry before antibody incubation.
- Methanol/ acetone fixation plus detergent permeabilization: After methanol/ acetone fixation and air-drying dip slide either in a solution containing 0.1-0.2% Triton X-100 in PBS or in 0.1% saponin in PBS for 1-5 min at room temperature (enhances accessibility of many cytoskeletal antigens).
- Air-drying of the section.
- Block with the serum of the species in which the secondary antibody was raised for 30 min.
- Incubation with 1st antibody 1 h at RT in moist chamber.
- Wash 3x with PBS.
- Incubation with appropriate fluorescent secondary antibody, 30-60 min at RT.
- Wash 3x with PBS.
- Immerse shortly into ethanol.
- Let air dry.
- Cover with mounting medium.
- For normal cell culture and methanol/acetone fixation of cells no precoating of glass coverslips is necessary.
- In case of formaldehyde fixation, cells should be grown on glass coverslips coated with 0.01% poly-L-lysine.
For fixation of lipid droplets within cells preferentially PBS containing 2% formaldehyde at RT for 10–20 min should be used. After fixation, the cells should be washed 3x for 5 min with PBS containing 50 mM NH4Cl to quench residual-free aldehyde groups. Thereafter, two permeabilization steps using PBS containing 0.2% saponin for 7–10 min at RT should be done prior to the application of primary antibodies. - For IF staining, methanol/acetone fixation is recommended: Cells grown on glass coverslips should be rinsed in PBS and fixed for 5 min in methanol followed by acetone (20 s), both pre-cooled at -20°C.
Briefly air dry the samples and rehydrate in PBS prior to the immunostaining procedure. After permeabilization with 0.2% Triton X-100 in PBS for 5 min at RT, wash cells several times in PBS.
In guinea pigs the antibody concentration in serum varies from 10 to 20 mg/ml; specific antibodies represent normally about 0.1-1% of total IgG. Total protein concentration varies from 40 to 65 mg/ml, with the main constituent (about 60%) being albumin.